13.3
Impact Factor
Theranostics 2022; 12(11):4851-4865. doi:10.7150/thno.69551 This issue Cite
Research Paper
Arterial cyclic stretch regulates Lamtor1 and promotes neointimal hyperplasia via circSlc8a1/miR-20a-5p axis in vein grafts
1. Institute of Mechanobiology & Medical Engineering, School of Life Sciences & Biotechnology, Shanghai Jiao Tong University, Shanghai, China.
2. Department of Pediatric Surgery, Xinhua Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai, 200092, China.
3. Department of Endocrinology and Metabolism, Shanghai Jiao Tong University Affiliated Sixth People's Hospital, Shanghai Diabetes Institute, Shanghai Clinical Center for Diabetes, Shanghai Key Laboratory of Diabetes Mellitus, Shanghai Key Clinical Center for Metabolic Disease, Shanghai, China.
Abstract
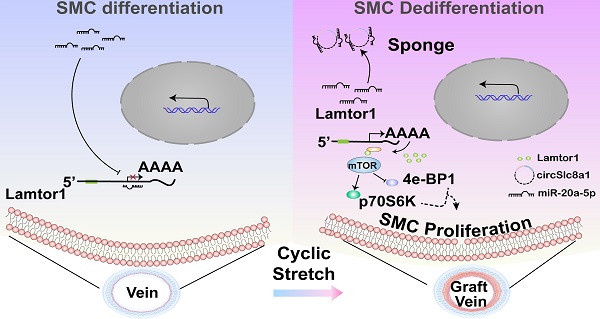
Rationale: Neointimal hyperplasia caused by dedifferentiation and proliferation of venous smooth muscle cells (SMCs) is the major challenge for restenosis after coronary artery bypass graft. Herein, we investigated the role of Lamtor1 in neointimal formation and the regulatory mechanism of non-coding RNA underlying this process.
Methods: Using a “cuff” model, veins were grafted into arterial system and Lamtor1 expression which was correlated with the activation of mTORC1 signaling and dedifferentiation of SMCs, were measured by Western blot. Whole transcriptome deep sequencing (RNA-seq) of the grafted veins combined with bioinformatic analysis identified highly conserved circSlc8a1 and its interaction with miR-20a-5p, which may target Lamtor1. CircSlc8a1 was biochemically characterized by Sanger sequencing and resistant to RNase R digestion. The cytoplasmic location of circSlc8a1 was shown by fluorescence in situ hybridization (FISH). RNA pull-down, luciferase assays and RNA immunoprecipitation (RIP) with Ago2 assays were used to identify the interaction circSlc8a1 with miR-20a-5p. Furthermore, arterial mechanical stretch (10% elongation) was applied in vitro.
Results: In vivo, Lamtor1 was significantly enhanced in grafted vein and activated mTORC1 signaling to promote dedifferentiation of SMCs. Arterial mechanical stretch (10% elongation) induced circSlc8a1 expression and positively regulated Lamtor1, activated mTORC1 and promoted SMC dedifferentiation and proliferation. Local injection of circSlc8a1 siRNA or SMC-specific Lamtor1 knockout mice prevented neointimal hyperplasia in vein grafts in vivo.
Conclusions: Our study reveals a novel mechanobiological mechanism underlying the dedifferentiation and proliferation of venous SMCs in neointimal hyperplasia. CircSlc81/miR-20a-5p/Lamtor1 axis induced by arterial cyclic stretch may be a potential clinical target that attenuates neointimal hyperplasia in grafted vessels.
Keywords: Lamtor1, circSlc8a1, mTORC1, cyclic stretch, vein graft
 Global reach, higher impact
Global reach, higher impact