Impact Factor
Theranostics 2022; 12(17):7390-7403. doi:10.7150/thno.73873 This issue Cite
Research Paper
Depletion of gut microbiota improves the therapeutic efficacy of cancer nanomedicine
1. Institute of Biomedical Sciences, Academia Sinica, Taipei 115, Taiwan.
2. Institute of Medical Genomics and Proteomics and Institute of Clinical Medicine, National Taiwan University College of Medicine, Taipei 100, Taiwan.
Abstract
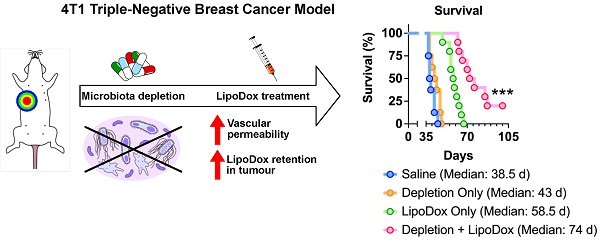
Rationale: Gut microbiota plays a crucial role in cancer development and treatment. Studies show that although the gut microbiota is able to promote tumor growth, its presence also improves the efficacy of cancer treatment such as immunotherapy. To date, understanding of the potential impact of the gut microbiota on other treatment modalities such as cancer nanomedicine is still limited. In this study, we aimed to establish the relationship between gut microbiota and cancer nanomedicine, which can potentially open a new path in cancer treatment that combines gut microbiota modulation along with nanotherapeutics.
Methods: Mice bearing 4T1 triple-negative breast cancer cells were subjected to gut microbiota modulation by antibiotics (ABX) treatment in the drinking water. Mice given normal water was used for control. The effects of ABX treatment towards gut bacteria was studied by RT-qPCR and 16S next generation sequencing of fecal samples. The mice were then subjected to liposomal doxorubicin (LipoDox) treatment and the amount of nanotherapeutics that accumulated in the tumors was quantified. For therapeutic efficacy, the mice were subjected to ABX treatment and given three injections of LipoDox or saline, while the tumor growth was monitored throughout.
Results: Analysis of fecal bacterial content showed that ABX treatment resulted in depletion of gut microbiota. Quantification of LipoDox content revealed significantly increased accumulation in ABX tumor compared to control. Compared to LipoDox treatment alone, we found that combined gut microbiota depletion and LipoDox treatment resulted in augmented long-term anti-tumor efficacy and significantly improved median survival compared to LipoDox only (control vs ABX = 58.5 vs 74 days, p = 0.0002, n = 10 for both groups), with two mice surviving until the end of the experimental end point without experiencing relapse. We also identified the increase in vascular permeability of ABX-treated tumors correlated to for improved therapeutic efficacy and outcome.
Conclusion: We showed that gut microbiota depletion led to enhanced tumor vascular permeability, which allowed a larger amount of LipoDox nanoparticles to accumulate in the tumor, leading to better long-term effects. Our results suggest that gut microbiota modulation may be exploited in combination with available nanomedicine-based therapeutics to improve cancer diagnosis, therapeutic efficacy and outcome.
Keywords: Gut microbiota, triple-negative breast cancer, nanomedicine, LipoDox, vascular permeability
 Global reach, higher impact
Global reach, higher impact