Impact Factor
Theranostics 2023; 13(4):1355-1369. doi:10.7150/thno.80544 This issue Cite
Research Paper
The delta subunit of the GABAA receptor is necessary for the GPT2-promoted breast cancer metastasis
1. Hongqiao International Institute of Medicine, Tongren Hospital; Basic Medical Institute; Key Laboratory of Cell Differentiation and Apoptosis of the Chinese Ministry of Education, Shanghai Jiao Tong University School of Medicine
2. Department of Laboratory Medicine, Shanghai General Hospital Jiading Branch, Shanghai
3. College of Basic Medical Sciences, Dalian Medical University
4. Department of Clinic Laboratory, Tongren Hospital, Shanghai Jiao Tong University School of Medicine
5. Department of Oncology, Shanghai East Hospital, Tongji University School of Medicine
#: Authors contributed equally to this article.
Abstract
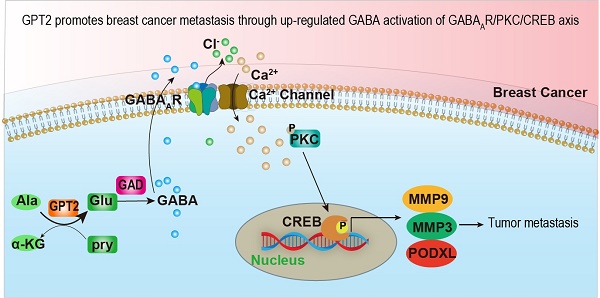
Objectives: Glutamic pyruvate transaminase (GPT2) catalyzes the reversible transamination between alanine and α-ketoglutarate (α-KG) to generate pyruvate and glutamate during cellular glutamine catabolism. The glutamate could be further converted to γ-aminobutyric acid (GABA). However, the role of GPT2 in tumor metastasis remains unclear.
Methods: The wound healing and transwell assays were carried out to analyze breast cancer cell migration and invasion in vitro. Gene ontology analysis was utilized following RNA-sequencing to discover the associated molecule function. The mass spectrometry analysis following phosphoprotein enrichment was performed to discover the associated transcription factors. Most importantly, both the tail vein model and Mammary gland conditional Gpt2-/- spontaneous tumor mouse models were used to evaluate the effect of GPT2 on breast cancer metastasis in vivo.
Results: GPT2 overexpression increases the content of GABA and promotes breast cancer metastasis by activating GABAA receptors. The delta subunit GABRD is necessary for the GPT2/GABA-induced breast cancer metastasis in xenograft and transgenic mouse models. Gpt2 knockout reduces the lung metastasis of the genetic Gpt2-/- breast cancer in mice and prolongs the overall survival of tumor burden mice. Mechanistically, GPT2-induced GABAA receptor activation increases Ca2+ influx by turning on its associated calcium channel, and the surged intracellular calcium triggers the PKC-CREB pathway activation. The activated transcription factor CREB accelerates breast cancer metastasis by upregulating metastasis-related gene expressions, such as PODXL, MMP3, and MMP9.
Conclusion: In summary, this study demonstrates that GPT2 promotes breast cancer metastasis through up-regulated GABA activation of GABAAR-PKC-CREB signaling, suggesting it is a potential target for breast cancer therapy.
Keywords: GPT2, breast cancer, GABA, GABA receptor, metastasis, calcium
 Global reach, higher impact
Global reach, higher impact