13.3
Impact Factor
Theranostics 2023; 13(11):3794-3813. doi:10.7150/thno.82975 This issue Cite
Research Paper
Ferritin light chain promotes the reprogramming of glioma immune microenvironment and facilitates glioma progression
1. Department of Neurosurgery, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, 450052, China.
2. The Application Center for Precision Medicine, Academy of Medical Science, Zhengzhou, 450052, China.
3. Henan Key Laboratory of Child Brain Injury and Henan Clinical Research Center for Child Neurological Disorders, Institute of Neuroscience and The Third Affiliated Hospital of Zhengzhou University, Zhengzhou, 450052, China.
4. Cancer Center, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, 450052, China.
5. National Engineering Laboratory for Internet Medical Systems and Applications, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, 450052, China.
6. Department of Hematology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, 450052, China.
7. Department of Endocrinology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, 450052, China.
8. Department of Pathology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, 450052, China.
#Authors contributed equally to this work.
Abstract
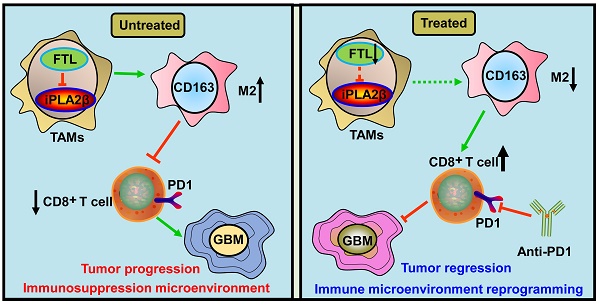
Background: Tumor-associated macrophages (TAMs), the most abundant non-tumor cell population in the glioma microenvironment, play a crucial role in immune evasion and immunotherapy resistance of glioblastoma (GBM). However, the regulatory mechanism of the immunosuppressive TME of GBM remains unclear.
Methods: Bioinformatics were used to analyse the potential role of ferritin light chain (FTL) in GBM immunology and explore the effects of FTL on the reprogramming of the GBM immune microenvironment and GBM progression.
Results: The FTL gene was found to be upregulated in TAMs of GBM at both the bulk and single-cell RNA-seq levels. FTL contributed to the protumor microenvironment by promoting M2 polarization in TAMs via inhibiting the expression of iPLA2β to facilitate the ferroptosis pathway. Inhibition of FTL in TAMs attenuated glioma angiogenesis, promoted the recruitment of T cells and sensitized glioma to anti-PD1 therapy.
Conclusion: Our study suggested that FTL promoted the development of an immunosuppressive TME by inducing M2 polarization in TAMs, and inhibition of FTL in TAMs reprogrammed the TME and sensitized glioma to anti-PD1 therapy, providing a new strategy for improving the therapeutic effect of anti-PD1.
Keywords: Glioblastoma, Ferritin light chain, Tumor-associated macrophages, macrophage polarization, PD1
 Global reach, higher impact
Global reach, higher impact