Impact Factor
Theranostics 2023; 13(13):4430-4448. doi:10.7150/thno.85369 This issue Cite
Research Paper
Secretome of senescent hepatic stellate cells favors malignant transformation from nonalcoholic steatohepatitis-fibrotic progression to hepatocellular carcinoma
1. Department of Medical Microbiology & Parasitology, MOE/NHC/CAMS Key Laboratory of Medical Molecular Virology, School of Basic Medical Sciences, Fudan University, Shanghai 200032, China.
2. Department of Gastroenterology, Shanghai Jing'an District Central Hospital, Fudan University, Shanghai 200040, China.
3. Joint Laboratory of Biomaterials and Translational Medicine, Puheng Technology Co., Ltd, Suzhou 215163, China.
4. Department of Gastroenterology & Hepatology, Zhongshan Hospital, Fudan University, Shanghai 200032, China.
5. Shanghai Institute of Liver Diseases, Fudan University Shanghai Medical College, Shanghai 200032, China.
Abstract
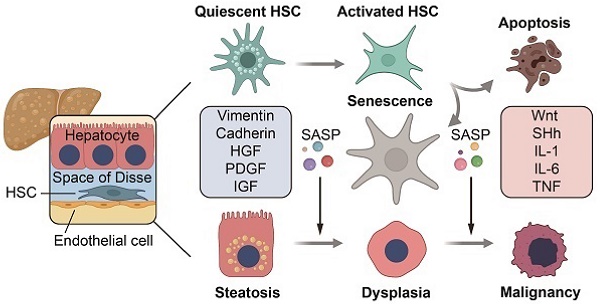
Background: Hepatic fibrosis is a premalignant lesion, and how injured hepatocytes transform into malignancy in a fibrotic microenvironment is poorly understood. Senescence is one of major fates of activated hepatic stellate cells (HSCs). Paucity of literature is available regarding the influence of senescent HSCs on behavior of steatotic hepatocytes.
Methods: Senescent HSCs were identified in a murine model of nonalcoholic steatohepatitis (NASH)-fibrosis-hepatocellular carcinoma (HCC) and human NASH-HCC specimens. Secretome of senescent HSCs was analyzed by label-free mass-spectrum (NanoRPLC-MS/MS) and verified quantitatively.
Results: Senescent HSCs were increased along with the progression from nonalcoholic fatty liver (NAFL), NASH to NASH-fibrosis, and reached a peak at the stage of advanced fibrosis and then decreased when hepatocellular dysplasia or HCC was developed. Critical components affecting proliferation, epithelial-mesenchymal transition (EMT) or migration were identified from secretome of senescent HSCs, and may activate morphogenic hedgehog or oncogenic Wnt signaling pathways to accelerate malignant transformation of steatotic or dysplastic hepatocytes. Primary hepatocytes stimulated with conditioned medium from senescent HSCs, in co-culture or co-cultured in 3D spheroids with senescent HSCs exhibited an enhanced proliferating or EMT profile.
Conclusion: Senescent HSCs secreted a characterized protein profile favoring malignant transformation of steatotic or dysplastic hepatocytes through activating morphogenic hedgehog or oncogenic Wnt signaling pathways in the progression from NASH to malignancy.
Keywords: Nonalcoholic steatohepatitis, Hepatocellular carcinoma, Hepatic stellate cell, Senescence, Senescence-associated secretary phenotype
 Global reach, higher impact
Global reach, higher impact