13.3
Impact Factor
Theranostics 2024; 14(3):924-939. doi:10.7150/thno.92193 This issue Cite
Research Paper
Macroscale pseudo-spheroids fabricated using methacrylated collagen-coated cells
1. Department of Precision Medicine, Sungkyunkwan University School of Medicine (SKKU-SOM) Suwon 16419, Republic of Korea.
2. Department of Biotechnology and Bioinformatics, Korea University, Sejong, Republic of Korea.
3. Biomedical Science and Engineering, Gwangju Institute of Science and Technology, Gwangju, Republic of Korea.
4. Institute of Quantum Biophysics, Department of Biophysics, Sungkyunkwan University, Suwon, Gyeonggi-do 16419, Republic of Korea.
5. Biomedical Institute for Convergence at SKKU (BICS), Sungkyunkwan University, Suwon 16419, Republic of Korea.
Abstract
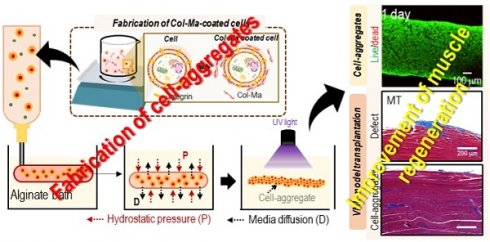
Rationale: Cell spheroids have shown great promise as tools for creating effective three-dimensional (3D) tissue models, facilitating tissue reconstruction and organoid development, due to their high cell density and efficient cellular interactions. However, a significant challenge persists in creating large-scale tissue structures with a 3D geometrical architecture using spheroids, due to the continual condensation and reorganization of cells and their environments.
Methods: The spherical cell aggregates (pseudo-cell spheroids) or macroscale cell aggregates were obtained by coating each adipose-derived stem cell (hASC) with methacrylated collagen (Col-Ma). Subsequently, the coated cells were printed into an alginate supporting bath and photocrosslinked through exposure to UV light. To assess the effectiveness of this procedure on regenerative potential, the generated cell aggregates were compared with conventional cell spheroids and bioprinted cell constructs using immunofluorescent staining and quantification of myogenic-related gene expressions. Moreover, the bioconstructs were implanted into a mouse model with volumetric muscle loss to further elucidate their regenerative and functional recovery properties.
Results: The use of Col-Ma as a cell-coating material enables the rapid and physical aggregation of cells within several hours, regardless of the cell type. Furthermore, Col-Ma-coated cell aggregates can provide relatively lower hypoxic conditions than cell spheroids fabricated using the hanging drop method owing to the thin porous Col-Ma layer coated on the cells. In addition, the resulting structures maintain their geometrical architecture following cell fusion and possess the potential for efficient scale-up and 3D complex shape formation, making them more suitable for clinical applications than conventional cell spheroids. Finally, the feasibility of the Col-Ma-coated cylindrical human adipose-derived stem cells aggregates was assessed through implantation in a mouse volumetric muscle loss model, showing a significantly higher regenerative ability of muscle tissue than the normally bioprinted cell construct.
Conclusion: Our newly proposed method has meaningful potential for various tissue engineering applications, supported by the improved cellular activities and efficient muscle regeneration observed in both in vitro and in vivo studies, and organ-chip models.
 Global reach, higher impact
Global reach, higher impact