Impact Factor
Theranostics 2024; 14(3):1260-1288. doi:10.7150/thno.89380 This issue Cite
Review
Understanding AAV vector immunogenicity: from particle to patient
1. Gene and Stem Cell Therapy Program Centenary Institute, The University of Sydney, NSW, Australia.
2. Faculty of Medicine and Health, The University of Sydney, NSW, Australia.
3. Pfizer Inc, Walton on the Hill, Surrey, UK.
4. Pfizer Australia, Sydney, NSW, Australia.
5. Pfizer Inc, New York, NY, USA.
6. Cell and Molecular Therapies, Royal Prince Alfred Hospital, Sydney, NSW, Australia.
Abstract
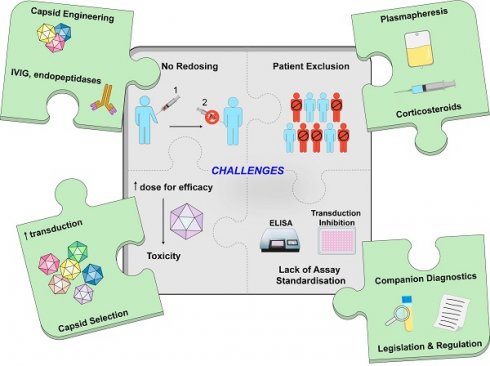
Gene therapy holds promise for patients with inherited monogenic disorders, cancer, and rare genetic diseases. Naturally occurring adeno-associated virus (AAV) offers a well-suited vehicle for clinical gene transfer due to its lack of significant clinical pathogenicity and amenability to be engineered to deliver therapeutic transgenes in a variety of cell types for long-term sustained expression. AAV has been bioengineered to produce recombinant AAV (rAAV) vectors for many gene therapies that are approved or in late-stage development. However, ongoing challenges hamper wider use of rAAV vector-mediated therapies. These include immunity against rAAV vectors, limited transgene packaging capacity, sub-optimal tissue transduction, potential risks of insertional mutagenesis and vector shedding. This review focuses on aspects of immunity against rAAV, mediated by anti-AAV neutralizing antibodies (NAbs) arising after natural exposure to AAVs or after rAAV vector administration. We provide an in-depth analysis of factors determining AAV seroprevalence and examine clinical approaches to managing anti-AAV NAbs pre- and post-vector administration. Methodologies used to quantify anti-AAV NAb levels and strategies to overcome pre-existing AAV immunity are also discussed. The broad adoption of rAAV vector-mediated gene therapies will require wider clinical appreciation of their current limitations and further research to mitigate their impact.
Keywords: neutralizing antibodies, humoral immunity, seroprevalence, patient exclusion, redosing
 Global reach, higher impact
Global reach, higher impact