Impact Factor
Theranostics 2018; 8(9):2435-2446. doi:10.7150/thno.22144 This issue Cite
Research Paper
Harmine enhances type H vessel formation and prevents bone loss in ovariectomized mice
1. Movement System Injury and Repair Research Center, Xiangya Hospital, Central South University, Changsha, Hunan 410008, China.
2. Hunan Key Laboratory of Organ Injury, Aging and Regenerative Medicine, Changsha, Hunan 410008, China.
3. Department of Sports Medicine, Xiangya Hospital, Central South University, Changsha, Hunan 410008, China.
4. Department of Orthopedics, Xiangya Hospital, Central South University, Changsha, Hunan 410008, China.
5. National Clinical Research Center for Geriatric Disorders, Xiangya Hospital, Central South University, Changsha, Hunan 410008, China.
6. Department of Spine Surgery, Xiangya Hospital, Central South University, Changsha, Hunan, 410008, China.
7. Department of Forensic Science, School of Basic Medical Sciences, Central South University, Changsha, Hunan 410013, China.
8. Department of Orthopaedic Surgery, Johns Hopkins University School of Medicine, Baltimore, Maryland 21205, USA
9. Department of Clinical Pharmacology, Xiangya Hospital, Central South University, Changsha, Hunan 410008, China.
10. The Second Xiangya Hospital, Central South University, Changsha, Hunan 410008, China.
11. China Orthopedic Regenerative Medicine Group (CORMed), Changsha, Hunan 410008, China.
* these authors contributed equally to this work.
Abstract
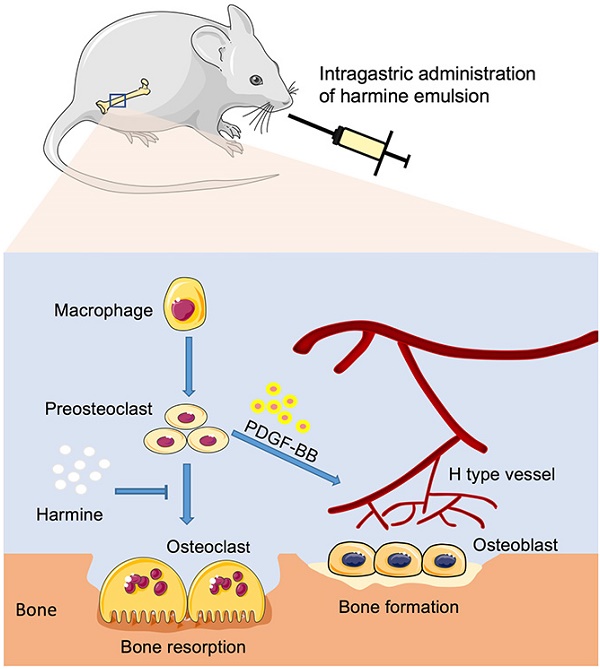
Recently, researchers identified a distinct vessel subtype called type H vessels that couple angiogenesis and osteogenesis. We previously found that type H vessels are reduced in ovariectomy (OVX)-induced osteoporotic mice, and preosteoclasts are able to secrete platelet-derived growth factor-BB (PDGF-BB) to stimulate type H vessel formation and thereby to promote osteogenesis. This study aimed to explore whether harmine, a β-carboline alkaloid, is capable of preventing bone loss in OVX mice by promoting preosteoclast PDGF-BB-induced type H vessel formation.
Methods: The impact of harmine on osteoclastogenesis of RANKL-stimulated RAW264.7 cells was verified by gene expression analysis and tartrate-resistant acid phosphatase (TRAP) staining. Enzyme-linked immunosorbent assay (ELISA) was conducted to test PDGF-BB production by preosteoclasts. A series of angiogenesis-related assays in vitro were performed to assess the pro-angiogenic effects of the conditioned media from RANKL-stimulated RAW264.7 cells treated with or without harmine. Meanwhile, the role of PDGF-BB in this process was determined. In vivo, OVX mice were intragastrically administrated with harmine emulsion or an equal volume of vehicle. 2 months later, bone samples were collected for µCT, histological, immunohistochemical and immunofluorescent analyses to evaluate bone mass, osteogenic and osteoclastic activities, as well as the numbers of type H vessels. Bone marrow PDGF-BB concentrations were assessed by ELISA.
Results: Exposure of RANKL-stimulated RAW264.7 cells to harmine enhanced the formation of preosteoclasts and the production of PDGF-BB. Harmine augmented the ability of RANKL-stimulated RAW264.7 cells to promote angiogenesis of endothelial cells, whereas the effect was blocked by PDGF-BB inhibition. In vivo, the oral administration of harmine emulsion to OVX mice resulted in enhanced trabecular bone mass and osteogenic responses, increased numbers of preosteoclasts, as well as reduced numbers of osteoclasts and fat cells. Moreover, OVX mice treated with harmine exhibited higher levels of bone marrow PDGF-BB and much more type H vessels in bone.
Conclusion: Harmine may exert bone-sparing effects by suppression of osteoclast formation and promotion of preosteoclast PDGF-BB-induced angiogenesis.
Keywords: harmine, preosteoclast, PDGF-BB, angiogenesis, osteogenesis
 Global reach, higher impact
Global reach, higher impact