Impact Factor
Theranostics 2019; 9(6):1634-1650. doi:10.7150/thno.30206 This issue Cite
Research Paper
Pseurotin A Inhibits Osteoclastogenesis and Prevents Ovariectomized-Induced Bone Loss by Suppressing Reactive Oxygen Species
1. School of Biomedical Sciences, University of Western Australia, Perth, WA 6009, Australia.
2. Department of Orthopedic Surgery, Sir Run Run Shaw Hospital, Medical College of Zhejiang University, Hangzhou, Zhejiang 310000, China
3. Key Laboratory of Musculoskeletal System Degeneration and Regeneration Translational Research of Zhejiang Province, Hangzhou, Zhejiang 310000, China
4. School of Physical Education and Sports Science, South China Normal University, Guangzhou, Guangdong 510631, China
5. Guangzhou University of Chinese Medicine, Guangzhou, Guangdong 510000, China
6. Department of Spine Surgery, Xiangya Hospital, Central South University, Changsha, Hunan 410008, China
7. Research Centre for Regenerative Medicine, Guangxi Medical University, Nanning, Guangxi 530021, China
8. Guangxi Key Laboratory of Regenerative Medicine, Guangxi Medical University, Nanning, Guangxi 530021, China
9. International Joint Laboratory on Regeneration of Bone and Soft Tissues, Guangxi Medical University, Nanning, Guangxi 530021, China
*Kai Chen and Pengcheng Qiu contributed equally to this work.
Abstract
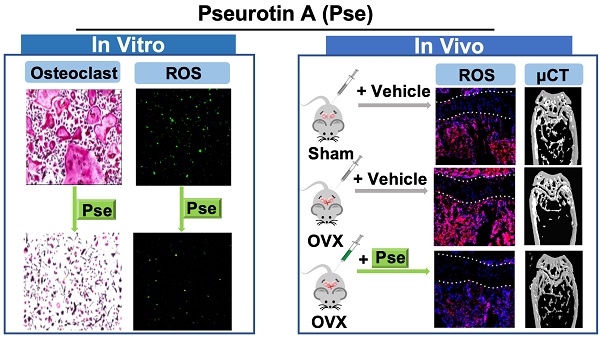
Rationale: Growing evidence indicates that intracellular reactive oxygen species (ROS) accumulation is a critical factor in the development of osteoporosis by triggering osteoclast formation and function. Pseurotin A (Pse) is a secondary metabolite isolated from Aspergillus fumigatus with antioxidant properties, recently shown to exhibit a wide range of potential therapeutic applications. However, its effects on osteoporosis remain unknown. This study aimed to explore whether Pse, by suppressing ROS level, is able to inhibit osteoclastogenesis and prevent the bone loss induced by estrogen-deficiency in ovariectomized (OVX) mice.
Methods: The effects of Pse on receptor activator of nuclear factor-κB (NF-κB) ligand (RANKL)-induced osteoclastogenesis and bone resorptive function were examined by tartrate resistant acid phosphatase (TRAcP) staining and hydroxyapatite resorption assay. 2',7'-dichlorodihydrofluorescein diacetate (H2DCFDA) was used to detect intracellular ROS production in vitro. Western blot assay was used to identify proteins associated with ROS generation and scavenging as well as ROS-mediated signaling cascades including mitogen-activated protein kinases (MAPKs), NF-κB pathways, and nuclear factor of activated T cells 1 (NFATc1) signaling. The expression of osteoclast-specific genes was assessed by qPCR. The in vivo potential of Pse was determined using an OVX mouse model administered with Pse or vehicle for 6 weeks. In vivo ROS production was assessed by intravenous injection of dihydroethidium (DHE) into OVX mice 24h prior to killing. After sacrifice, the bone samples were analyzed using micro-CT and histomorphometry to determine bone volume, osteoclast activity, and ROS level ex vivo.
Results: Pse was demonstrated to inhibit osteoclastogenesis and bone resorptive function in vitro, as well as the downregulation of osteoclast-specific genes including Acp5 (encoding TRAcP), Ctsk (encoding cathepsin K), and Mmp9 (encoding matrix metalloproteinase 9). Mechanistically, Pse suppressed intracellular ROS level by inhibiting RANKL-induced ROS production and enhancing ROS scavenging enzymes, subsequently suppressing MAPK pathway (ERK, P38, and JNK) and NF-κB pathways, leading to the inhibition of NFATc1 signaling. Micro-CT and histological data indicated that OVX procedure resulted in a significant bone loss, with dramatically increased the number of osteoclasts on the bone surface as well as increased ROS level in the bone marrow microenvironment; whereas Pse supplementation was capable of effectively preventing these OVX-induced changes.
Conclusion: Pse was demonstrated for the first time as a novel alternative therapy for osteoclast-related bone diseases such as osteoporosis through suppressing ROS level.
Keywords: Pseurotin A, reactive oxygen species, osteoclast, osteoporosis
 Global reach, higher impact
Global reach, higher impact