Impact Factor
Theranostics 2019; 9(15):4287-4307. doi:10.7150/thno.36077 This issue Cite
Research Paper
The role of Nav1.7 and methylglyoxal-mediated activation of TRPA1 in itch and hypoalgesia in a murine model of type 1 diabetes
1. Jiangsu Key Laboratory of Neuropsychiatric Diseases and the Second Affiliated Hospital of Soochow University, Suzhou 215004, P.R. China.
2. Institute of Neuroscience, Soochow University, Suzhou, 215021, P.R. China.
3. Departments of Anesthesiology, Duke University Medical Center, Durham, North Carolina, 27710, USA.
4. Department of Neurobiology, Duke University Medical Center, Durham, North Carolina, 27710, USA.
5. Jiangsu Key Laboratory of Preventive and Translational Medicine for Geriatric Diseases, Department of Nutrition and Food Hygiene, School of Public Health, Soochow University, Suzhou, 215021, P.R. China.
6. College of Life Sciences, Yanan University, Yanan, 716000, China
7. Present address: State Key Laboratory of Bio-organic and Natural Products Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, Shanghai, 200032, China
#These authors contributed equally to this study.
Abstract
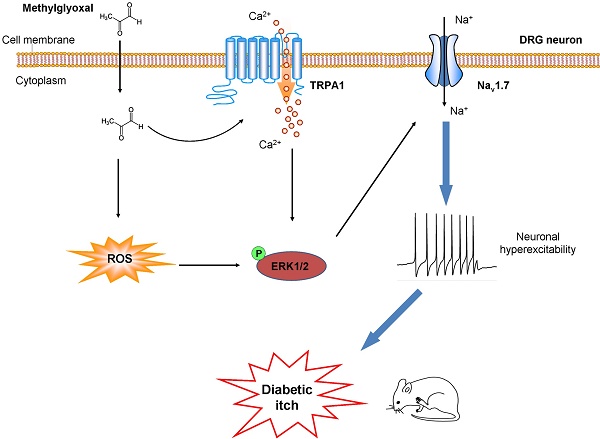
Methylglyoxal (MGO), an endogenous reactive carbonyl compound, plays a key role in the pathogenesis of diabetic neuropathy. The aim of this study is to investigate the role of MGO in diabetic itch and hypoalgesia, two common symptoms associated with diabetic neuropathy.
Methods: Scratching behavior, mechanical itch (alloknesis), and thermal hypoalgesia were quantified after intradermal (i.d.) injection of MGO in naïve mice or in diabetic mice induced by intraperitoneal (i.p.) injection of streptozotocin (STZ). Behavioral testing, patch-clamp recording, transgenic mice, and gene expression analysis were used to investigate the mechanisms underlying diabetic itch and hypoalgesia in mice.
Results: I.d. injection of MGO evoked dose-dependent scratching in normal mice. Addition of MGO directly activated transient receptor potential ankyrin 1 (TRPA1) to induce inward currents and calcium influx in dorsal root ganglia (DRG) neurons or in TRPA1-expressing HEK293 cells. Mechanical itch, but not spontaneous itch was developed in STZ-induced diabetic mice. Genetic ablation of Trpa1 (Trpa1-/-), pharmacological blockade of TRPA1 and Nav1.7, antioxidants, and mitogen-activated protein kinase kinase enzyme (MEK) inhibitor U0126 abrogated itch induced by MGO or in STZ-induced diabetic mice. Thermal hypoalgesia was induced by intrathecal (i.t.) injection of MGO or in STZ-induced diabetic mice, which was abolished by MGO scavengers, intrathecal injection of TRPA1 blockers, and in Trpa1-/- mice.
Conclusion: This study revealed that Nav1.7 and MGO-mediated activation of TRPA1 play key roles in itch and hypoalgesia in a murine model of type 1 diabetes. Thereby, we provide a novel potential therapeutic strategy for the treatment of itch and hypoalgesia induced by diabetic neuropathy.
Keywords: Itch, Hypoalgesia, Diabetes, TRPA1, Methylglyoxal
 Global reach, higher impact
Global reach, higher impact