Impact Factor
Theranostics 2020; 10(14):6467-6482. doi:10.7150/thno.43865 This issue Cite
Research Paper
MGAT3-mediated glycosylation of tetraspanin CD82 at asparagine 157 suppresses ovarian cancer metastasis by inhibiting the integrin signaling pathway
1. State Key Laboratory of Medicinal Chemical Biology, School of Medicine, Nankai University, Tianjin, China
2. Department of Cardiac Development and Remodeling, Max-Planck-Institute for Heart and Lung Research, Bad Nauheim, Germany
3. Department of Cancer Biology, Wake Forest University School of Medicine, Winston-Salem, NC, USA
Abstract
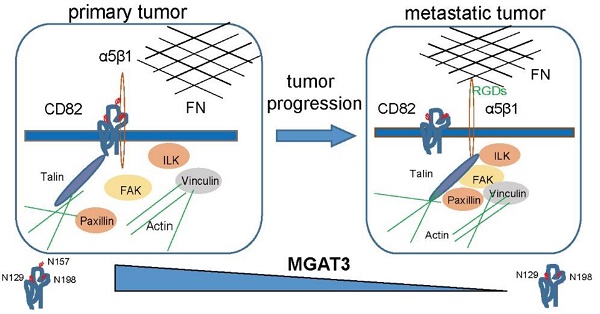
Background: Tetraspanins constitute a family of transmembrane spanning proteins that function mainly by organizing the plasma membrane into micro-domains. CD82, a member of tetraspanins, is a potent inhibitor of cancer metastasis in numerous malignancies. CD82 is a highly glycosylated protein, however, it is still unknown whether and how this post-translational modification affects CD82 function and cancer metastasis.
Methods: The glycosylation of CD82 profiles are checked in the paired human ovarian primary and metastatic cancer tissues. The functional studies on the various glycosylation sites of CD82 are performed in vitro and in vivo.
Results: We demonstrate that CD82 glycosylation at Asn157 is necessary for CD82-mediated inhibition of ovarian cancer cells migration and metastasis in vitro and in vivo. Mechanistically, we discover that CD82 glycosylation is pivotal to disrupt integrin α5β1-mediated cellular adhesion to the abundant extracellular matrix protein fibronectin. Thereby the glycosylated CD82 inhibits the integrin signaling pathway responsible for the induction of the cytoskeleton rearrangements required for cellular migration. Furthermore, we reveal that the glycosyltransferase MGAT3 is responsible for CD82 glycosylation in ovarian cancer cells. Metastatic ovarian cancers express reduced levels of MGAT3 which in turn may result in impaired CD82 glycosylation.
Conclusions: Our work implicates a pathway for ovarian cancers metastasis regulation via MGAT3 mediated glycosylation of tetraspanin CD82 at asparagine 157.
Keywords: Ovarian cancer, Metastasis, Glycosylation, CD82, Exosomes
 Global reach, higher impact
Global reach, higher impact