Impact Factor
Theranostics 2022; 12(4):1486-1499. doi:10.7150/thno.66119 This issue Cite
Research Paper
CD40- and 41BB-specific antibody fusion proteins with PDL1 blockade-restricted agonism
1. Division of Molecular Internal Medicine, Department of Internal Medicine II, University Hospital Würzburg, Würzburg, Germany.
2. Department of Hematology, University of Groningen, University Medical Center Groningen, Groningen, The Netherlands.
3. Medizinische Klinik und Poliklinik II, Universitätsklinikum Würzburg, Würzburg, Germany.
4. Interdisciplinary Center for Clinical Research Laboratory, Department of Internal Medicine II, University Hospital Würzburg, Würzburg, Germany.
Abstract
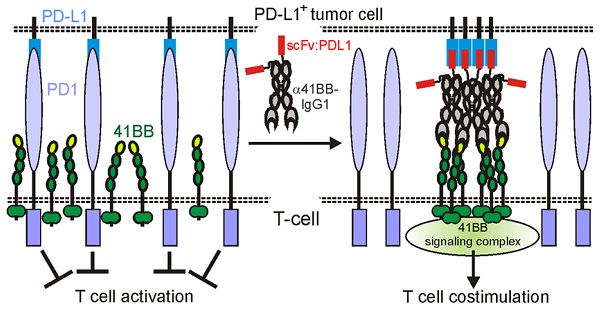
Background: A strategy to broaden the applicability of checkpoint inhibitors is the combined use with antibodies targeting the immune stimulatory receptors CD40 and 41BB. However, the use of anti-CD40 and anti-41BB antibodies as agonists is problematic in two ways. First, anti-CD40 and anti-41BB antibodies need plasma membrane-associated presentation by FcγR binding to exert robust agonism but this obviously limits their immune stimulatory efficacy by triggering ADCC, CDC or anti-inflammatory FcγRIIb activities. Second, off tumor activation of CD40 and 41BB may cause dose limiting systemic inflammation.
Methods: To overcome the FcγR-dependency of anti-41BB and anti-CD40 antibodies, we genetically fused such antibodies with a PDL1-specific blocking scFv as anchoring domain to enable FcγR-independent plasma membrane-associated presentation of anti-CD40- and anti-41BB antibodies. By help of GpL-tagged variants of the resulting bispecific antibodies, binding to their molecular targets was evaluated by help of cellular binding studies. Membrane PDL1-restricted engagement of CD40 and 41BB but also inhibition of PDL1-induced PD1 activation were evaluated in coculture assays with PDL1-expressing tumor cell lines and 41BB, CD40 and PD1 responsible cell lines or T-cells.
Results: The binding properties of the bispecific antibody fusion proteins remained largely unchanged compared to their parental molecules. Upon anchoring to membrane PDL1, the bispecific antibody fusion proteins activated CD40/41BB signaling as efficient as the parental anti-CD40/anti-41BB antibodies when bound to FcγRs or cells expressing membrane-bound CD40L/41BBL. PD1 inhibition remained intact and the anti-41BB fusion protein thus showed PDL1-restricted costimulation of T-cells activated in vitro with anti-CD3 or a BiTe.
Conclusions: Targeting of anti-CD40 and anti-41BB fusion proteins to membrane PDL1 with a blocking PDL1 scFv links PD1-PDL1 checkpoint blockade intrinsically with engagement of CD40 or 41BB.
Keywords: 41BB, bispecific antibody, CD40, PDL1, TNFRSF
 Global reach, higher impact
Global reach, higher impact